July 5, 2019
The Foundation for Biomedical Research and Innovation at Kobe (FBRI), Tokyo Electron Ltd., and Shimadzu Corporation together analyzed the culture media of human-induced pluripotent stem cells (iPSCs) and embryonic stem cells (ESCs) over time and successfully identified specific metabolites secreted during undifferentiated state maintenance and early differentiation that represent markers indicating the differentiation status of these cells. This discovery allows the maintenance of an undifferentiated state and differentiation to be determined without the need to destroy the cells. In the future, these markers may be used in process control for real time analysis of cell status in ongoing cultures. Shimadzu’s LCMS-8050 ultra fast liquid chromatograph mass spectrometer was used in this research. The results of this research that was conducted in partnership between these three organizations were published in Science Signaling on June 26.
1. Background
Human iPSCs and ESCs are called pluripotent stem cells, with both cell lines having the ability to proliferate almost indefinitely while maintaining an undifferentiated*1 state (potential for self-renewal) and to differentiate*2 into various cell and tissue types when given a differentiation stimulus (differentiation potential). The combination of these two abilities allows regenerative medicine to use pluripotent stem cells to produce differentiated cells at numbers required for transplantation.
Although undifferentiated cells are cultured in media that maintain their undifferentiated state, undifferentiated cells will naturally start to differentiate if culture media is not replaced at the proper frequency, or if culture is continued after proliferation of too many cells. When differentiation is induced in a mixture of undifferentiated cells and naturally differentiated cells, it is safe to assume that 100 % of cells will not differentiate into the desired cell type. Transplantation of an incomplete mixture of differentiated cells is unlikely to produce the proper intended effect. For this reason, a simple method of monitoring the differentiation status of cells that analyzes culture media without destroying cells is essential for safe and effective practice of cell therapy utilizing iPSCs and ESCs. A simple method of culture monitoring requires markers that indicate the differentiation status of cells in culture.
2. Research Method and Results
Using Shimadzu’s LCMS-8050 ultra fast liquid chromatograph mass spectrometer, Foundation for Biomedical Research and Innovation at Kobe, Tokyo Electron Ltd., and Shimadzu Corporation analyzed the culture media of undifferentiated and early-differentiating iPSCs and ESCs over time. This analysis led to the discovery that kynurenine is secreted into culture media when cells maintain an undifferentiated state, and 2-aminoadipic acid is secreted into culture media when cells begin to differentiate. These two factors can be used as markers to determine the differentiation status of cells through culture media analysis without cell destruction.
More specifically, undifferentiated cells take up the essential amino acid tryptophan and use it to form kynurenine, then by binding with AhR*3 in the cell cytoplasm, the complex of kynurenine and AhR is believed to be transported into the cell nucleus where it induces the expression of genes that maintain an undifferentiated cellular state. By contrast, when cells receive a differentiation stimulus, in order to escape this undifferentiated state, a catabolic pathway is initiated that breaks down kynurenine and excretes it outside the cell. This kynurenine metabolism results in the extracellular elimination of 2-aminoadipic acid, which is the final product of this catabolic pathway. Detection of 2-aminoadipic acid in culture media therefore indicates early differentiation.
3. Ripple Effects and Future Plans
The identification of differentiation factors secreted into culture media can be utilized as a simple and noninvasive method of implementing quality control for iPSC cultures in real time. Utilization of this method will help improve the safety of medical transplantation of iPSC-derived cells. In-process monitoring of cells by culture media analysis is shown to be an effective means of quality control.
Article Title and Authors
Kynurenine signaling through the aryl hydrocarbon receptor maintains the undifferentiated state of human embryonic stem cells
Takako Yamamoto, Kunitada Hatabayashi, Mao Arita, Nobuyuki Yajima, Chiemi Takenaka, Takashi Suzuki, Masatoshi Takahashi, Yasuhiro Oshima, Keisuke Hara, Kenichi Kagawa, Shin Kawamata
Glossary
| *1 Undifferentiated | : | A cell state in which the cell has yet to change into a specific cell type |
| *2 Differentiation | : | The process or action of differentiating into one of a variety of different cell or tissue types |
| *3 AhR | : | Aryl hydrocarbon receptor |
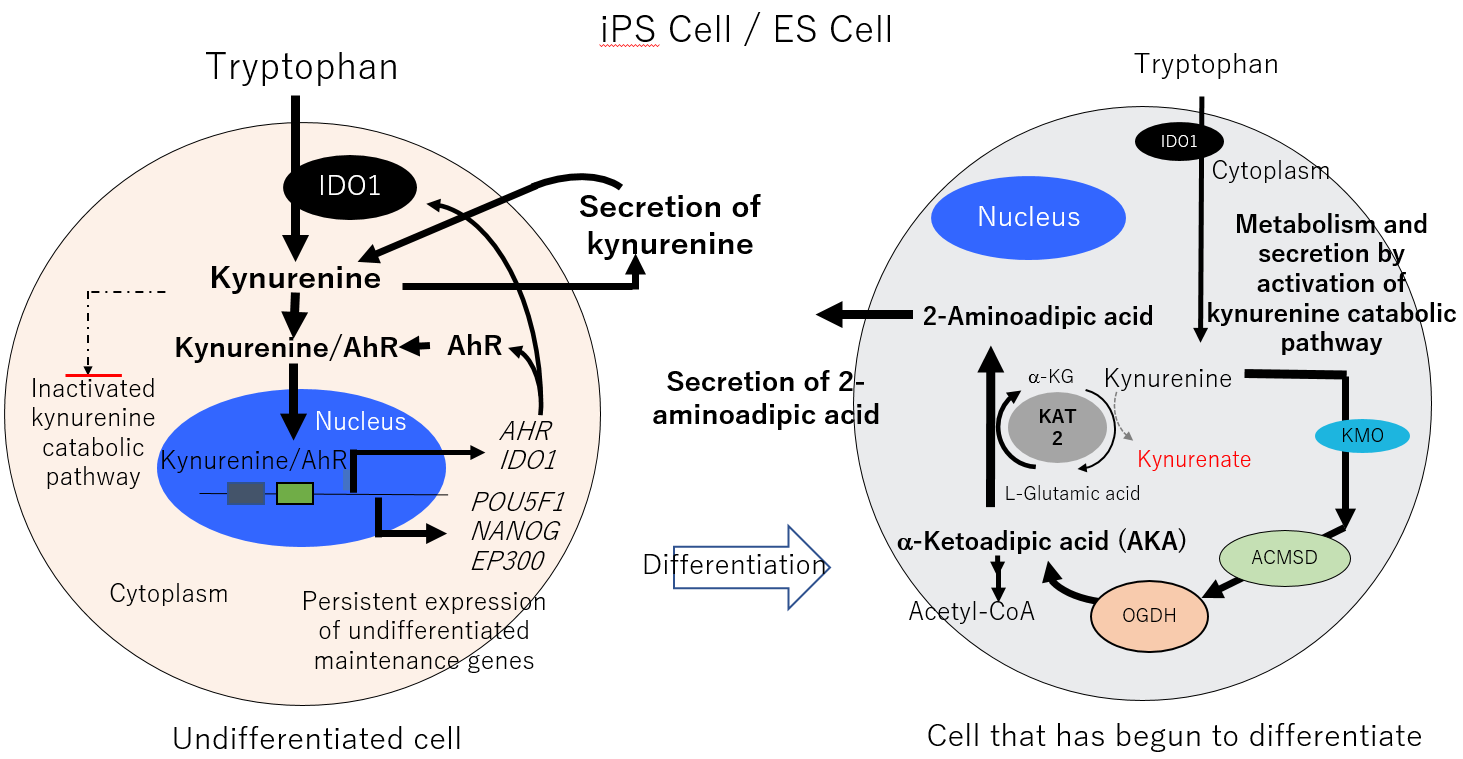
Fig. Mechanism of Differentiation

Photograph: LCMS-8050 Ultra Fast Liquid Chromatograph Mass Spectrometer





